Together with Veeva Vault Clinical Suite, Veeva is the first and only company providing solutions that help sponsors, sites, and patients share and view information during a clinical trial. In addition to Vault Site Connect, Veeva today announced MyVeeva, a multichannel patient portal for virtual visits and patient-centric trials. Vault Site Connect is part of the Veeva Clinical Network, a set of solutions that brings together sponsors, sites, and patients to accelerate clinical research. “Now the industry can leverage the same innovative Veeva Vault technology to drive greater efficiency and productivity during trials.” “Veeva Vault Site Connect will eliminate the many portal interfaces, manual handoffs, and multiple steps it takes to share data with sponsors during a trial,” said Tonya Yarbrough, director, Vanderbilt Institute for Clinical and Translational Research at Vanderbilt University Medical Center. Now sites can focus less on administrative tasks and responding to information requests and more on execution and treating patients, improving overall study quality. With Vault Site Connect, sponsors can improve site engagement by reducing the time and effort in exchanging clinical information. Sponsors can easily collect input from sites on study feasibility to spend less time coordinating surveys accelerate document exchange and reconciliation between eTMF and eISF quickly distribute safety letters to notify global sites and regulators of an adverse event and get real-time information on the status of patients from enrollment through treatment – all with one application. Vault Site Connect seamlessly automates information flow across trial partners, processes, and systems. 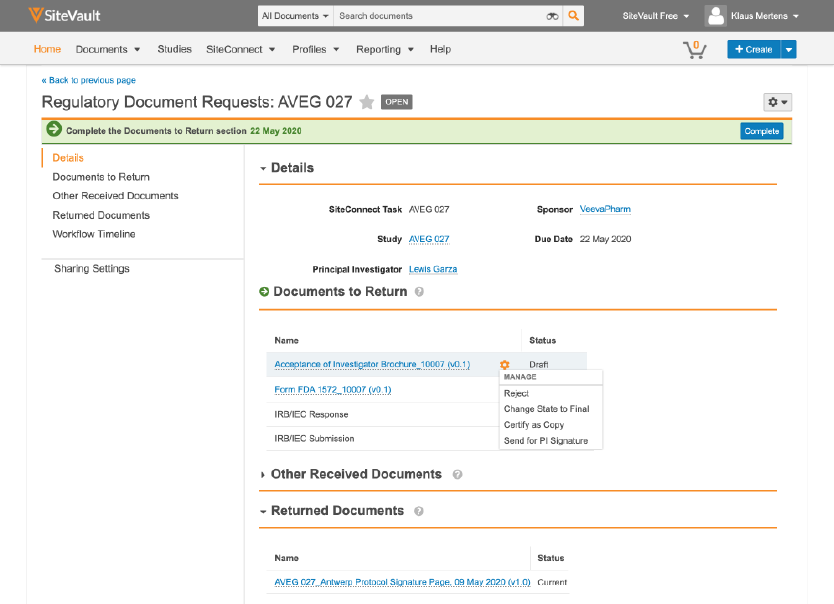
“Veeva Vault Site Connect will transform how sponsors and sites work together and help get studies up and running much faster.” site management and monitoring at AstraZeneca. August 3, 2021.“There is a significant opportunity to speed information exchange throughout the clinical trial process,” said Doug Schantz, member of SCRS leadership council and former head of U.S. ^ "Veeva Acquires Learnaboutgmp, a Leading GxP eLearning Content Provider | Veeva".^ "Veeva Acquires Physicians World | Veeva".^ "Veeva to Acquire Crossix, the Leader in Privacy-Safe Patient Data and Analytics – Crossix".^ "Veeva scoops up rival Zinc Ahead to create mega-content management platform" (Press release).^ "Veeva Becomes First Public Company to Convert to a Public Benefit Corporation".Class A Common Stock (VEEV) Quote & Summary Data". Annual Report fiscal year ending Janu(Form 10-K)". In 2021, Veeva acquired Learnaboutgmp LLC, which provides compliance training services for life sciences organizations. In 2019, Veeva acquired Physicians World LLC, a pharmaceutical company that provides business process outsourcing services. In 2019, Veeva acquired Crossix, a privacy-safe patient data and analytics company. In 2015, Veeva acquired Zinc Ahead, a content management software company. This made it the first publicly-traded company to convert to a public benefit corporation. 
On February 1, 2021, Veeva became a public benefit corporation. As of May 5, 2022, it has a market capitalization of US$27.5 billion. It works with software as a service (SaaS) in the life-science industry. Headquartered in Pleasanton, California, it was founded in 2007 by Peter Gassner and Matt Wallach. is an American cloud-computing company focused on pharmaceutical and life sciences industry applications. Physicians World LLC, Crossix Solutions, Zinc Ahead, Learnaboutgmp LLC
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
